Do genetic variations influence the effects of dietary or supplemental fatty acid intake on health outcomes?
The effects of dietary and supplemental omega-3 and omega-6 fatty acids are varied, and some of this variation may be explained by the genetics of fatty acid metabolism. In particular, the fatty acid desaturase (FADS) enzyme has genetic variation that may influence the rate at which omega-3 and omega-6 precursors are metabolized downstream into long-chain polyunsaturated fatty acids.
Abstract
The effects of dietary and supplemental omega-3 and omega-6 fatty acids are varied, and some of this variation may be explained by the genetics of fatty acid metabolism. In particular, the fatty acid desaturase (FADS) enzyme has genetic variation that may influence the rate at which n-3 and n-6 precursors are metabolized downstream into long-chain polyunsaturated fatty acids. A systematic review of the last 6 years of research shows that dietary fatty acid intake seems to be most influenced by FADS genetic variation in terms of cardiovascular outcomes and lipid markers. The effect of fatty acid intake on other health outcomes may not be as strongly influenced by FADS variation, though some exceptions do exist. This review will also outline how clinicians can estimate FADS activity and variation using fatty acid analysis.
Introduction
Nutrigenomics is a budding field of medicine that takes into account the patient’s individual genetic makeup when considering nutritional recommendations and supplementation. Certainly, much attention has been given to the role of omega-3 (n-3) fatty acids in health in recent years. Now, nutrigenomics brings to light the possible role of genetic variation in fatty acid metabolism. This paper reviews the recent evidence and seeks to answer the question: Do FADS genotype variations influence the effects of dietary or supplemental fatty acid intake on health outcomes?
The synthesis of long-chain polyunsaturated fatty acids (LC-PUFAs) such as eicosapentoic acid (EPA) and docosahexanoic acid (DHA) involves multiple steps featuring desaturase and elongase enzymes. Delta-6 desaturase converts linoleic acid (LA), an n-6 fatty acid, and alpha-linolenic acid (ALA), an n-3 fatty acid, into gamma-linoleic acid (GLA) and stearidonic acid (SDA), respectively. Delta-5 desaturase converts dihomo-gamma-linoleic acid (DGLA) into arachidonic acid (AA), while this same enzyme converts the n-3 fatty acid pathway downstream into EPA (Figure). These desaturase enzymes are encoded by the FADS gene cluster found on chromosome 11.1 Fatty acid desaturase 1 (FADS1) corresponds to delta-5 desaturase; fatty acid desaturase 2 (FADS2) corresponds to delta-6 desaturase. Polymorphisms in the FADS1 and FADS2 genes are directly associated with abnormal functioning of these desaturase enzymes, leading to alterations in the synthesis of LC-PUFAs. Studies indicate that carriers of the FADS1 or FADS2 minor allele have a reduced activity of delta-5 desaturase and delta-6 desaturase, respectively. This means that, in carriers of the minor alleles, the substrates of the affected enzyme would accumulate, while the end products, such as EPA and DHA, would be deficient.2 Conversely, the FADS1 and FADS2 major alleles are associated with increased delta-5 desaturase and delta-6 desaturase activity, respectively. In carriers of a major allele, we would expect lower levels of the enzymes’ substrates and higher levels of the enzymes’ end products. It is important to keep in mind that EPA and DHA are LC-PUFAs that are substrates of the desaturase enzymes. Arachidonic acid (AA) is synthesized by delta-5 desaturase, so an increase in delta-5 desaturase will create an increase in AA. This paper is a systematic review of the last 6 years of evidence regarding the role that FADS1 and FADS2 polymorphisms play in health—in particular, the evidence surrounding how varied FADS genotypes modify the behavior of n-3 and n-6 fatty acid intake, either through diet or supplementation, in the body. The review focuses specifically on cardiovascular outcomes but also reviews evidence for other health outcomes.
Methods
Literature review of PubMed, AltHealthWatch, Allied and Complementary Medicine Database (AMED), Annual Reviews, the Cumulative Index to Nursing and Allied Health Literature (CINAHL), Cochrane Library, JSTOR Health Sciences, and Science Direct. Search terms included the following: FADS1; FADS2; FADS1 supplementation; FADS2 supplementation; FADS intake; and FADS polymorphism diet. The search yielded 275 papers. Those were further narrowed down by restricting the review to papers published in 2010 or later and papers that evaluated the association between dietary or supplemental n-3 or n-6 fatty acid intake with outcomes for different FADS genotypes. Health outcomes included primary clinical outcomes, biomarkers, and essential fatty acid (EFA) status in plasma, serum, or red blood cells (RBCs). Multiple study designs were allowed, including randomized trials and observational studies. There was no restriction on sample size or dose of n-3 or n-6 fatty acid supplementation. A total of 22 papers met the inclusion criteria and were analyzed in this review. The quality of the evidence was categorized as high, moderate, or low as described in Table 1. No meta-analysis was performed due to the varied types of studies and outcomes included in this review.
Results and Discussion
Since 2010, a number of studies have evaluated the effect of genetic interactions with dietary n-3 and n-6 fatty acid intake on cardiovascular outcomes. A 2010 study by Lu et al evaluated the effect of dietary n-3 or n-6 fatty acid intake with FADS variations on total cholesterol, high-density lipoprotein cholesterol (HDL-C), and non-HDL-C levels in 3,575 participants.1 The mean intake of n-3 polyunsaturated fatty acids was 0.53% of total energy, while the mean n-6 polyunsaturated fatty acid intake was 5.47% of total energy. The patients with the FADS1 major allele, indicating increased delta-5 desaturase activity, who also had the highest n-3 fatty acid intake had the highest levels of total cholesterol and non-HDL-C levels. Levels of HDL-C were highest in those who also had the major allele of FADS1 and who had the highest level of n-6 fatty acid intake.
In 2012, Hellstrand et al evaluated 4,635 people using a dietary history to evaluate n-3 and n-6 fatty acid intake. The minor allele of FADS1, indicating slow delta-5 desaturase activity, was associated with lower low-density lipoprotein cholesterol (LDL-C) levels in those who ate the lowest amount of fish and n-3 LC-PUFAs. This same effect was not found in those homozygous for the major allele. Additionally, a higher ratio of ALA to LA (ALA/LA ratio) in the diet was significantly associated with higher HDL-C levels in those carrying the minor allele (and therefore with slower delta-5 desaturase activity).3 In another study, Liu et al evaluated 444 patients with existing coronary artery disease (CAD) and 538 healthy control subjects. Participants were tested for their FADS genotype and their associated EPA and DHA dietary intakes. In this study, the minor allele was associated with an increased risk of having CAD in those with the lowest EPA intake but not in those with the highest EPA intake [odds ratio (OR): 1.36]. The minor allele carriers with the lowest DHA intake also had an increased risk of CAD.4 The same risk was not found in the major allele carriers.
In a 2013 study by Zhu et al, 210 Han Chinese patients were evaluated for their FADS genotype, lipid status, and dietary fatty acid intake. The study found that the minor allele was associated with reduced levels of arachidonic acid (AA) and higher levels of DGLA. These minor allele carriers had higher levels of triglycerides but no significant changes in LDL-C or total cholesterol levels. The minor allele was only associated with lower HDL-C levels when LA status was low. The minor allele was associated with low HDL-C and high triglycerides in those with lowest ALA intake.5 In 2012 Hellstrand and colleagues published another study. It evaluated the effects FADS1 variations on the association between dietary fatty acid intake and the risk of cardiovascular disease in 24,032 participants without cardiovascular disease or diabetes, over the course of 14 years. Investigators found a borderline interaction between the dietary ALA/LA ratio and the FADS1 genotype on the incidence of cardiovascular disease. Those with the minor allele, and therefore the lowest delta-5 desaturase activity, had an inverse relationship between ALA/LA ratio in the diet and the risk of cardiovascular disease. In other words, the higher the ALA intake and/or the lower the LA intake, the lower the risk of cardiovascular disease in the minor allele carriers. There was also a significant association between ALA intake and the FADS1 genotype on the incidence of ischemic stroke in the homozygous minor allele genotype.6
Standl et al studied 2,006 10-year-old children to evaluate associations between the FADS genotype and total cholesterol, HDL-C, LDL-C , and triglyceride levels. In this study, higher n-3 fatty acid intake was associated with higher HDL-C levels and lower triglyceride levels in those with the FADS1 variations. The study did not find any association between lipid level, FADS genotype, and dietary n-6 fatty acid intake.7 A smaller evaluation of 36 hyperlipidemic patients compared fatty acid status of patients consuming a diet rich in ALA in the form of flax seed oil, a diet rich in canola oil, or a Western diet. The patients were evaluated for both their FADS genotype and their plasma fatty acid levels. The minor allele carriers at baseline had lower levels of EPA and AA, and lower EPA to ALA (EPA/ALA) and AA to LA (AA/LA) ratios regardless of the diet they followed. The study also revealed that those minor allele homozygotes who then followed the flax seed-enriched diet (with the highest ALA dietary intake, reaching 10% of total energy) had an increase in plasma EPA levels beyond even that of the major allele carriers in the Western diet group, thus compensating for their weakened baseline EPA status. This study also confirmed previous findings that those with FADS variations who made the dietary changes did not have any significant alteration in DHA plasma levels. Lastly, the study found the lowest levels of AA in those minor allele carriers who were in the flax seed group.8
One study of 573 European adolescents evaluated whether or not dietary ALA and LA intake affects the association of FADS variations on blood lipids. High LA intake was determined to be anything over 9.4 g/day and high ALA intake was anything over 1.4 g/day. The study found that the FADS minor allele was associated with lower total cholesterol and non-HDL-C in those with the highest ALA dietary intake (around 1.8 g/day).9
Regarding cardiovascular outcomes, the quality of evidence in these studies is inconsistent, with certain studies having a large sample size and others a very low sample size. Additionally, the type of n-3 fatty acid intake or n-6 fatty acid intake was not standardized across the studies. However, despite inconsistent quality of evidence, the studies since 2010 have consistently demonstrated that FADS genetic variations, leading to varied delta-5 and delta-6 desaturase enzyme activity, influence the effect of dietary n-3 and n-6 fatty acids on cardiovascular disease markers and outcomes. When looking at the comprehensive list of studies, we find that increased n-3 fatty acid intake, typically in the form of ALA, is associated with increased HDL-C levels, reduced cardiovascular disease and stroke incidence, lower triglyceride levels, lower total cholesterol levels, and lower non-HDL-C levels in those with the slowest desaturase activity (minor allele FADS variants). The 2010 study by Lu et al found that higher n-3 fatty acid intake along with increased delta-5 desaturase activity in major allele variants was associated with increased total cholesterol and non-HDL-C levels.1 When the delta-5 desaturase enzyme is working at an increased activity level in major allele variants, and when those patients also ingest high levels of n-6 fatty acids, this study found elevated HDL-C. Interestingly, patients who consumed fewer dietary n-3 fatty acids, paired with slower delta-5 desaturase activity, had lower levels of LDL-C, increased risk of coronary artery disease, increased triglycerides, and lower total cholesterol levels. These same minor allele variant carriers with slower delta-5 desaturase activity also had lower HDL-C levels when they consumed fewer n-6 fatty acids.
Do FADS variations influence the effect of supplemental fatty acid intake on cardiovascular outcomes?
In the second phase of this systematic review, we evaluated the evidence regarding the effects of FADS variations on cardiovascular markers with n-3 fatty acid supplementation (in the form of fish oil capsules) to determine if there were similar patterns to those observed with dietary n-3 consumption. Overall, the evidence does not point to a consistent effect of fish oil supplementation on cardiovascular outcomes or markers in those with different FADS genotypes.
In 2014, Alsaleh et al evaluated the effect of FADS variation in 310 patients randomized to receive a placebo or a fish oil supplement. Members of the supplement group received either 0.45 g, 0.9 g, or 1.8 g EPA + DHA once a day for 12 months. Those supplemented with the highest dose of fish oil who were also the minor allele carriers for FADS1 had a 21.6% reduction in triglyceride levels after the intervention, compared to those homozygous for the major allele with the highest delta-5 desaturase activity. The homozygous major allele carriers had a 3.5% reduction in triglyceride levels after supplementation at the 1.8 g/day fish oil dose.10 This finding is in contrast to an earlier study that evaluated the effects of 3.0 g/day of fish oil (1.9 g/day EPA + 1.1 g/day DHA) on plasma lipid profiles in 208 subjects for 6 weeks. The supplement significantly improved triglyceride levels but there was no difference based on FADS genotype. Interestingly, 28.8% had no change in triglyceride levels after supplementation regardless of FADS genotype, possibly pointing to the role of other genetic influences beyond FADS genotype.11 One very small study of 12 young adults aged 18 to 25 evaluated the effects of 1.8 g/day of EPA+DHA fish oil supplementation for 12 weeks. The FADS1 variation was evaluated along with triglycerides, total cholesterol, LDL-C, HDL-C, cholesterol to HDL-C ratio, high sensitivity C-reactive protein (hs-CRP), and glycemic markers at regular intervals during the intervention period and the washout period. While there were no significant differences between the major and minor allele carriers in any of the cardiometabolic markers, the minor allele carriers had a significantly greater percent increase in RBC EPA levels during the supplementation period compared to the major allele carriers.12
Some studies have indicated that fish oil supplementation may have an impact on the activity level of desaturase enzyme activity regardless of FADS genotype. A 2012 study evaluated the effects of a 6-week supplementation with 5 g fish oil per day, providing 1.9 to 2.2 g/day EPA plus 1.1 g/day DHA. The study found that the supplementation increased the activity of delta-5 desaturase and decreased the activity of delta-6 desaturase. The triglyceride, insulin, total cholesterol, and LDL-C levels were inversely associated with the activity of delta-5 desaturase and positively associated with the level of delta-6 desaturase activity after the supplementation. The minor allele carriers, however, still had lower delta-5 desaturase activity even after 6 weeks of fish oil supplementation. Ultimately, there did not seem to be a differential effect on cardiometabolic markers based on FADS variation with the fish oil intervention.13 A similar result was found in a 2013 study of 310 healthy patients who were given 0.45 g, 0.9 g, or 1.8 g per day EPA+DHA for 6 months. Plasma lipids and desaturase enzyme activity was monitored in patients with FADS variations. This study also found that the fish oil supplements were associated with increased delta-5 desaturase activity and lowered delta-6 desaturase activity at all doses. The minor allele carriers had significantly lower delta-5 desaturase activity at baseline, as was expected, which subsequently significantly increased after supplementation (P=4.0x10-9).14
The evidence from recent studies evaluating the effects of fish oil supplementation on cardiovascular outcomes and markers according to FADS genotypes has not been as consistent as the evidence of the effects of dietary fatty acid intake. Fish oil supplementation seems to lower triglyceride levels regardless of FADS genotype, and it also seems to have an effect on levels of desaturase activity. Fish oil supplementation does not appear to affect plasma lipids in different ways based on FADS genotype.
According to the criteria in Table 1, 66% of studies evaluating cardiovascular outcomes or cardiovascular biomarkers were of moderate to high quality of evidence, while 34% were low to moderate quality. While the quality of the evidence is inconsistent, a number of trends have been noted. In those with low fatty acid desaturase enzyme activity who also eat a high amount of n-3 fatty acids, there is a pattern of increased HDL-C, lowered cardiovascular disease (CVD) and stroke incidence, lower triglyceride levels, lower total cholesterol levels, and lower non-HDL-C levels. Also, in those with low fatty acid desaturase enzyme activity who eat the lowest amount of n-3 fatty acids, there is a pattern for lower LDL-C and total cholesterol levels, but also increased risk of coronary artery disease and increased triglycerides. In those with low fatty acid desaturase enzyme activity who eat low n-6 fatty acid amounts, there is a pattern towards lower HDL-C levels. The relationship between fatty acid intake, FADS variations, and cholesterol levels is a complex one, but it is related to the amount and availability of polyunsaturated fatty acids with more than 4 double bonds in the body.1
When the desaturase enzymes are working slower, as occurs in those with FADS minor alleles, the substrates for those enzymes are elevated and the LC-PUFAs, which are the end products of desaturase enzymes, are reduced. The LC-PUFAs with over 4 double bonds normally stimulate peroxisome proliferator-activator receptors (PPARs). Stimulation of PPARs modifies the genetic expression of lipoproteins.1 For example, the stimulation of PPAR-alpha by LC-PUFAs modulates levels of HDL-C and triglycerides and regulates the genetic expression of lipoprotein lipase and apolipoproteins A-I, A-II, and C-III.6 This may explain why low desaturase activity, and therefore lower LC-PUFA availability, tends to lead to lower levels of lipoproteins in those who eat little n-3 or n-6 fatty acids. This is especially true for HDL-C levels. Another explanation is related to the idea that n-3 fatty acids and n-6 fatty acids compete for desaturase enzyme activity. When a person eats a high n-6 fatty acid content, linoleic acid accumulates especially in those with lower desaturase activity. This saturates the enzymes with n-6 fatty acid substrates, therefore inhibiting n-3 fatty acid metabolism into its LC-PUFAs and thus inhibiting the ability of LC-PUFA to increase HDL-C.15 This was confirmed by one study indicating that the minor allele carriers with the highest n-6 fatty acid intake had lower HDL-C levels.1 Furthermore, those with low desaturase activity who eat large amounts of alpha linolenic acid (ALA) will accumulate ALA. Large amounts of ALA may have positive effects on the risks of CVD, thus indicating a benefit of an ALA-rich diet in minor allele carriers at risk for CVD.7 The increased availability of ALA in the diet will flood the desaturase enzymes with ALA, thus increasing the availability of EPA and decreasing the availability of AA. This was confirmed by a 2013 study that found higher EPA levels in those with the lowest desaturase activity who increased their ALA intake to 10% of total calories compared to those consuming a baseline diet who had the highest desaturase activity.9 This indicates that a very large amount of ALA consumption can compensate for sluggish desaturase activity and lead to high EPA levels. Additionally, those with low desaturase enzyme activity with an increased concentration of precursor fatty acids, like ALA, may have increased membrane fluidity and reduced LDL-C levels.8 This was confirmed by a study of adolescents ingesting 1.8 g/day of ALA who had lowest desaturase activity. The adolescents who consumed 1.8 g/day of ALA had lower non-HDL-C cholesterol levels than those eating less ALA each day.10
In those with high desaturase activity, an increase in n-3 fatty acid dietary intake was associated with increased total cholesterol and non-HDL-C levels. Increased n-6 dietary fatty acid intake in those with increased desaturase activity also led to elevated HDL-C levels. Possible mechanisms have been discussed in the literature. One explanation is that a high dietary n-3 fatty acid intake paired with the highest desaturase enzyme activity may suppress the uptake of apolipoprotein B-containing particles, like very low-density lipoprotein cholesterol (VLDL-C) and LDL-C, thus leaving them elevated in the blood.1
However, the association between dietary fatty acid intakes, FADS variations, and plasma lipids may not only be explained by these mechanisms. Al-hilal and colleagues concluded that “lipids are distal phenotypes that are influenced by many genes and environmental factors in addition to desaturase activity.”16 We may not be able to explain the mechanism for the effects of FADS variations on lipid profiles in those with different fatty acid intakes because there may not be a linear explanation. These authors do conclude, however, that “some contribution [of FADS genotypes] is likely.”
Do FADS variations influence the effect of dietary or supplemental fatty acid intake on pregnancy or breastfeeding?
In addition to cardiovascular outcomes, the effects of FADS variations and dietary fatty acid intakes have also been evaluated in pregnant women and breastfeeding infants. A 2015 study by Yeates et al evaluated FADS1 and FADS2 variations along with dietary fish intake and its effect on Bayley Scales of Infant Development scores at 20 or 30 months. All 1,622 mothers in the study consumed 8.5 to 9.0 fish-containing meals per week. In these high fish eaters, the FADS1 and FADS2 homozygous minor alleles were associated with 20% lower AA levels and a 42% higher ratio of LA to AA (LA/AA ratio) in maternal serum. Overall, those with the minor alleles had higher levels of LA and ALA, and higher ALA to DHA ratios than those with major alleles. However, there was no association of the FADS variations with the infants’ Bayley scale scores, the primary outcome. There was a trend towards improved psychomotor development in infants of mothers with the minor allele for FADS1 and FADS2, but this trend was not statistically significant. One significant limitation of this study was the lack of a control group, as all women were high fish eaters.16 In 2013, Rizzi et al studied 534 twins to evaluate the relationship between breastfeeding and FADS gene variations on intelligence quotient (IQ). The study found a trend towards a positive effect of breastfeeding on IQ scores in children with the FADS major allele.17 FADS variations may possibly play a role in pregnancy duration and birth weight. Another study evaluated 2,622 mother-infant pairs and collected data on maternal DHA intake, pregnancy duration, birth weight, and FADS1 status. The results showed that women in the 75th percentile for DHA intake had longer pregnancies (+0.7 days) and heavier infants (+28 g) than women with lower DHA intake. Those with highest AA intake had shorter pregnancies (-0.6 days) than women in the lowest 25th percentile of AA intake. Those homozygous for the FADS1 minor allele had shorter pregnancies (-2 days) and lighter infants (-140 g) than those homozygous for the major allele. Those women in the 75th percentile for DHA intake who also were homozygous for the FADS1 minor allele had heavier infants (+226 g) than those with low DHA dietary intake. The FADS status of the fetus did not seem to affect these results.18
Two additional studies met our inclusion criteria, but did not evaluate a primary clinical outcome. Instead, these studies evaluated associations between DHA intake and DHA status in either the breast milk or RBCs in those with different FADS genotypes. One study (Moltó-Puigmartí et al) evaluated the effect of FADS variants and maternal fish and fish oil intake on the content of DHA in breast milk. At baseline, DHA was lower in homozygous minor allele FADS genotypes compared to homozygous major allele genotypes. Levels of DHA increased in the breast milk with increasing fish and fish oil intake in the major allele carriers, but the DHA content remained lower in those with the minor allele. While this is an interesting study, it fails to evaluate a significant primary clinical outcome.19 While DHA supplementation may not increase breast milk DHA content in minor allele carriers, one study did find that supplementation with DHA increases DHA status in the RBCs in pregnant women regardless of FADS genotype. This study of 205 pregnant women evaluated the relationship of FADS variations with DHA and AA status before and after the administration of either 600 mg of marine algal DHA supplementation or placebo once a day in the last 2 trimesters of pregnancy. The study found that all FADS genotypes had an increased DHA status in RBCs, and that members of the DHA-supplemented group who were homozygous for the FADS minor allele had lower AA levels.20 Limitations of this study include its failure to evaluate any direct clinical outcome.
In summary, the evidence has shown that in a very small number of studies, increased fish intake in those with lower desaturase enzyme activity may contribute to lower levels of AA in serum and RBCs and increased infant birth weight. Those with the highest AA intake may have a longer duration of pregnancy. There was also a trend towards improved psychomotor development in infants of minor allele mothers who had the highest fish intake. This trend may be due to the lower maternal levels of AA, and the authors conclude that the maternal balance of AA and DHA may influence neurodevelopment in infants.16 Generally it appears that consuming higher amounts of DHA while pregnant may reduce AA levels especially in those with the minor allele. It also appears that women with the minor allele may have lower levels of DHA in breast milk than those with the major allele. The study by Moltó-Puigmartí et al showed that women with the slowest desaturase activity who attempted to compensate with the highest amount of fish and fish oil intake still had lower breast milk DHA levels despite elevated plasma DHA levels. The authors surmise that FADS minor allele carriers may also have alterations in the ability to incorporate DHA into the milk, but this effect needs to be studied in further trials.19 This is clinically useful information. Ultimately, however, there is not enough high-quality evidence to make any definitive recommendations.
Do FADS variations influence the effect of dietary or supplemental fatty acid intake on other miscellaneous health outcomes?
The relationship between dietary fatty acid intake and FADS variations has also been explored to a lesser degree since 2010 in regards to other health outcomes. In 2013, Porenta et al evaluated the effects of diet on fatty acid status in the colonic mucosa of 108 patients at an increased risk for colon cancer. Participants followed either an n-3 fatty acid-rich Mediterranean diet or the Healthy Eating Diet, which is higher in n-6 fatty acids, for 6 months.21 The Mediterranean diet included 2 n-3 fatty acid rich-foods per week, with 6 servings of grains and 7 to 9 servings of fruits and vegetables per day. The Healthy Eating Diet, based on the recommended diet by Healthy People 2010, included 2 servings of fruit, 3 servings of vegetables, and 6 servings of grains per day, with less than 10% of the total calories from saturated fat and less than 30% from total fat. After the 6-month intervention period, those with FADS major alleles who ate the Mediterranean diet had 16% lower AA levels in the colonic mucosa than those who followed the Healthy Eating Diet. Members of the Healthy Eating Diet group who had FADS major alleles had higher colonic AA levels after 6 months. Though this study did not evaluate a primary clinical outcome, the results may be useful. In 2011, Standl et al evaluated the prevalence of atopy and both food-specific and inhalant-specific IgE levels in 10-year-old children. The study evaluated dietary fatty acid intake along with FADS genotype. The results showed that daily margarine intake was significantly and positively associated with asthma in those homozygous for the FADS major allele. There was also a nonsignificant trend towards an increased risk of allergic rhinitis (hay fever) in the homozygous major allele group who had a higher n-3 to n-6 fatty acid intake ratio.22 Lastly, a 2013 study evaluated the effects of 6 weeks of 5 g fish oil (1.9-2.2 g/d EPA + 1.1 g/d DHA) per day in 210 overweight subjects. Markers for glycemic control were evaluated as the primary outcome. Those with the FADS2 minor allele had increased fasting glucose after the trial period.23
Overall, 80% of the studies on miscellaneous health outcomes were of moderate-quality evidence, while 20% had low-quality evidence, based on the criteria described in Table 1. The combined evidence for recommendations on dietary fatty acid intake and other outcomes in relation to FADS variations is low due to both the failure to evaluate primary outcomes and small sample sizes. General conclusions that can be drawn include the association of a high n-3 fatty acid diet with less production of AA. An increase in n-3 fatty acids in the diet leads to less production of AA because the n-3 fatty acids flood the desaturase enzymes, which leads to EPA production and less AA production.5 This same mechanism may explain the finding of increased asthma rates in children with the highest margarine intake with the highest desaturase activity. The more n-6 fatty acid substrates available from the margarine-rich diet combined with high desaturase activity will lead to the highest AA levels, which will enhance the activity of cyclooxygenase and lipoxygenase for the synthesis of generally pro-inflammatory eicosanoids.9
Clinical Implications
The results of this literature review are summarized in Table 2. Ultimately, it appears that FADS variations may dictate the effects that dietary intake of n-3 and n-6 fatty acids have in the body. It appears that this effect is more potent for dietary fatty acid intake compared to supplementation with fatty acids, such as in fish oil supplements. Clinically evaluating a patient’s FADS genotype may be useful given that HDL-C and total cholesterol levels seem to respond, in certain studies, to n-3 fatty acid intake in an opposite fashion in those with low versus high desaturase activity. Those with low desaturase activity and a high dietary n-3 fatty acid intake will have a higher ALA status, which may prove to be clinically beneficial for patients at risk for heart disease. High n-6 fatty acid intake may raise or lower HDL-C levels depending on desaturase activity and FADS status. Testing for FADS polymorphisms themselves is not readily available at this time, but many of the studies reviewed here have utilized fatty acid ratios to assess FADS status. Delta-5 desaturase activity, and therefore FADS1 status, can be assessed by the AA to DGLA (AA/DGLA) ratio. Delta-6 desaturase activity, and therefore FADS2 status, can be assessed by the GLA to LA (GL/LA) ratio. This is an easy way for clinicians to predict FADS1 or FADS2 variation.
The effect of fish oil supplementation was not as pronounced as the effect of dietary fatty acid intakes. The one effect that was consistently found was an elevation in delta-5 desaturase activity and reduction in delta-6 desaturase activity with fish oil supplementation. Supplementation with fish oil may exhibit feedback inhibition of delta-6 desaturase through transcriptional suppression.24 The increased delta-5 desaturase activity may reflect a true increase in enzymatic activity, but it may also reflect a lower amount of DGLA, which is used to assess delta-5 desaturase activity. If delta-6 desaturase is suppressed, DGLA would be suppressed, which is used to calculate delta-5 desaturase activity. However, one study did find that modest n-3 LC-PUFA supplementation induced altered DNA methylation of the FADS enzyme genes.25 Therefore, if clinicians find aberrations in delta desaturase enzyme activity, fish oil or algal oil supplementation may modify the activity of these enzymes.
Lastly, a number of studies consistently demonstrate that DHA levels are altered by dietary intake and not by FADS status. There is limited biosynthesis of DHA in vivo, regardless of the patient’s FADS status, so there is a need for exogenous DHA consumption to increase DHA status, reflecting little genetic influence.9 Ultimately, clinicians may find it useful to utilize fatty acid ratios to assess desaturase enzyme activity in order to make recommendations about fatty acid intake.
Table 1. Criteria for Quality of Evidence
| Quality Categories | High | Moderate | Low |
| Design | Randomized, Intervention | Prospective | Retrospective |
| Control Group | Yes | Yes | No |
| Outcome | Clinical outcome | Clinical biomarker | EFA status |
| Adjustment for Confounding Factors | Yes; at least 3 factors | Yes; at least 1 factor | No |
Abbreviation: EFA, essential fatty acid.
Table 2. Summary of Clinical Outcomes/Clinical Biomarkers
| Dietary Fatty Acid Intake | Low Desaturase Activity | High Desaturase Activity |
| High n-3 fatty acids | +HDL-C levels -CVD, stroke incidence -TG levels -TC levels -non-HDL-C levels +infant birth weight -DHA breast milk content | +TC levels +non-HDL-C levels |
| High n-6 fatty acids | -HDL-C levels | +HDL-C levels +asthma rates in childhood |
| Low n-3 fatty acids | -LDL-C levels +CAD risk +TG levels -TC levels -DHA breast milk content | |
| Low n-6 fatty acids | -HDL-C levels | |
Key: +high, -low. Desaturase activity indicated by GLA/LA ratio and/or AA/DGLA ratio; high ratios indicate high activity, low ratios indicate low activity.
Figure
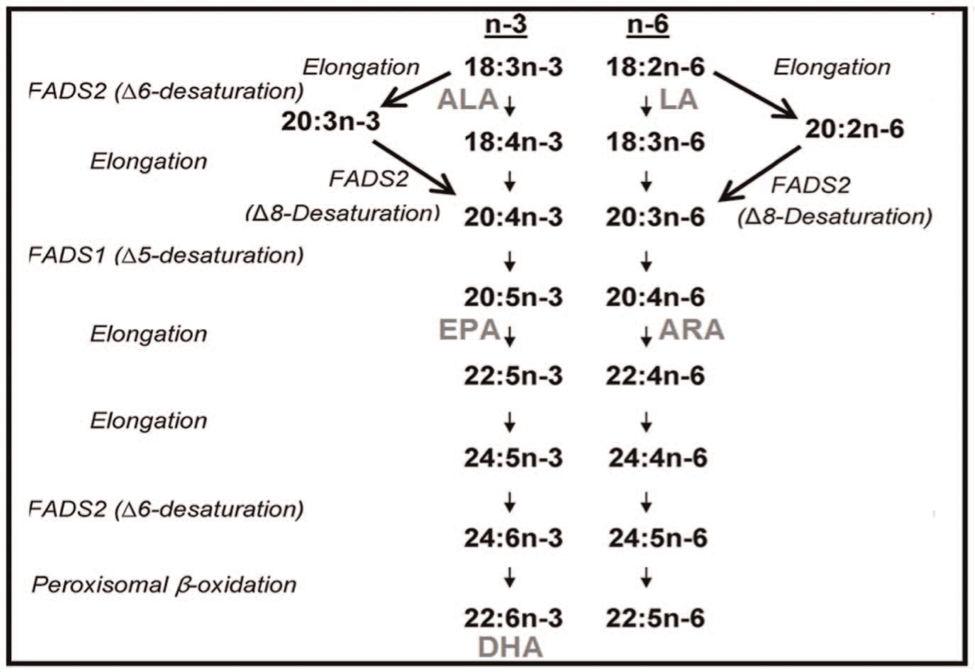
From Rizzi et al, 2013.17







